Posts Tagged ‘embryo recipient’
The Embryo Bank Dilemma: Reviewing the Issues, Historical Perspectives and Offering Potential Solutions

Craig R. Sweet, M.D
Reproductive Endocrinologist
Practice and Medical Director
Embryo Donation International
Introduction
In my last blog, “Why Creating ‘McEmbryos’ is Just Plain Wrong,” I wrote about my concerns regarding the creation of an embryo bank at a California clinic. In this follow-up segment, I want to re-state the issues, discuss the past history of embryo banking in the U.S., provide a list of recently written thoughtful blogs on the topic, offer possible solutions to the dilemma and discuss where we should go from here.
A “Reader’s Digest” version
California Conceptions (CC), as outlined in Alan Zarembo’s L.A. Times article apparently combined donor eggs with donor sperm and divided the resulting embryos among a number of embryo recipients. This process is commonly called a “split or shared donor/donor cycle” but was called “embryo donation” by CC. Any embryos remaining, after the recipients received their allotment, would be cryopreserved and owned by CC.
The road was paved with good intentions
I feel that CC was really trying to offer a cost-affective alternative for patients and that its true intent was to keep the size of its embryo bank as small as possible. Even with good intentions, however, it is quite likely that the embryo bank will grow. In addition, to sanction the creation of a small embryo bank will almost certainly result in the creation of larger embryo banks across the country. These banked embryos for commercial use are what I called “McEmbryos.” There also needs to be a clear distinction between embryo banking for commercial use and the process of banking one’s own embryos (i.e., collecting through multiple IVF retrievals) to be used by individuals to build their families in the future.
I still have three main concerns:
- I do not feel that embryo banks are appropriate and could result in a plethora of unintended consequences.
- I feel that corporations, businesses or physician practices should not own embryos.
- Lastly, the process of a “split or shared donor/donor cycle” should be called “embryo creation” or, at the very least, not called embryo donation.
This has happened before
An article by Gina Kolata in the New York Times in 1997 revealed that “ready-made embryos” were already being made for “adoption.” Columbia-Presbyterian and Reproductive Biology Associates were named in the article as providing “premade” embryos to patients. According to the article, most of the embryos were created when donor egg recipients backed out of the process, but the egg donors still underwent the egg retrieval; their subsequent retrieved donor oocytes were combined with donor sperm. Lori B. Andrews, a professor of law at Chicago-Kent College of Law, was quoted as having concerns about the supermarket approach to embryos while the clinicians thought it wasteful to not retrieve and fertilize the donor oocytes if the egg donors were ready for the retrieval.
In 2007, Center for Genetics and Society Senior Fellow and UC Hastings Law Professor Osagie Obasogie wrote an op-ed for the Boston Globe about a Texas center that had created an embryo bank. He was concerned about the “Wal-Martization” of human embryos, a phrase similar to my “McEmbryos.”
In November of 2012, in response to the N.Y. Times article, Jessica Cussins of the Center for Genetics and Society wrote an excellent blog on the topic, also following-up on the 2007 article by Professor Obasogie. The Texas center was eventually closed and was the subject of an FDA investigation, which eventually found that the creation of an embryo bank did not fall under FDA jurisdiction. John Robertson, Esq., wrote an excellent commentary on the topic in the Bioethics Forum in that same year.
Embryo banks have come and gone, garnering media attention and criticism and I believe it is finally time to set some ethical standards of care about them.
How New York decided to handle the embryo bank issue
 Almost five years ago, the state of New York issued regulations for tissue banks and nontransplant anatomic banks, addressing the potential of creating embryo banks:
Almost five years ago, the state of New York issued regulations for tissue banks and nontransplant anatomic banks, addressing the potential of creating embryo banks:
Embryos shall not be created for donation by fertilizing donor oocytes with donor semen, except at the request of a specific patient who intends to use such embryos for her own treatment. [NYS 52-8.7(h)]
Embryos were not to be created to store in embryo banks but only created at the behest of a specific patient and subsequently owned by that patient. Simply modifying the statement above to include “… such embryos for his/her own treatment,” would address the issue adequately, with the sentence potentially used by various organizations as they hopefully set ethical standards of care.
Potential consequences to the creation of an embryo bank
I have been called an alarmist by some for bringing up what I feel are the following potential dangers of having embryos banks in the U.S:
- If a small embryo bank is allowed to flourish, then large embryo banks will most certainly follow.
- Poorly designed and reactive legislation may be created on the state or national level as there may be further calls to regulate what are perceived to be “unregulated IVF facilities.”
- “Personhood” advocates may become further emboldened to win personhood for the embryos to protect them from becoming “McEmbryos.”
I don’t think these unintended consequences are that farfetched and need to be considered carefully should embryo banks continue unchecked.
My reluctant decision to come forward
 About the last thing I wanted to do was to comment on another reproductive endocrine practice comprised of caring staff members dedicated to the care of their patients. I have been criticized for taking such a stand and accused of doing this purely for competitive reasons. In reality, I have been working with the American Society for Reproductive Medicine’s (ASRM) and the Society for Assisted Reproductive Technologies, (SART) since October of 2011, trying to elicit a set of guidelines prior to the writing of my blog. I far preferred to stay out of the limelight and let the “powers-that-be” decide what should be done next. When the L.A. Times article was published, it de-emphasized the ethical issues and potential unintended consequences of the CC embryo banking practice, so I felt I had no choice but to bring the topic up front and center.
About the last thing I wanted to do was to comment on another reproductive endocrine practice comprised of caring staff members dedicated to the care of their patients. I have been criticized for taking such a stand and accused of doing this purely for competitive reasons. In reality, I have been working with the American Society for Reproductive Medicine’s (ASRM) and the Society for Assisted Reproductive Technologies, (SART) since October of 2011, trying to elicit a set of guidelines prior to the writing of my blog. I far preferred to stay out of the limelight and let the “powers-that-be” decide what should be done next. When the L.A. Times article was published, it de-emphasized the ethical issues and potential unintended consequences of the CC embryo banking practice, so I felt I had no choice but to bring the topic up front and center.
Others responded to the discussion
Several other infertility professionals discussed the ethical issue in articles or blogs in the weeks following the L.A. Times piece. Excluding those that simply summarized the situation, I listed below what I think are some of the better blogs:
Supporting embryo banking
- Marni Soupcoff, Esq., “Marni Soupcoff on the sale of fertilized embryos: How much for the blastocyst in the window?”
Neutral to embryo banking
- Julie Shapiro, Esq., “Custom Made or Off The Rack?”
- Carole C. Wegner, Ph.D., “Embryos for Donation: Where are the ethical boundaries?“
- Elizabeth Swire Falker, Esq., “The Bizarre World of Embryo Banking. Where My Motherhood and Morality Meet”
Against embryo banking
- Andrew Vorzimer, Esq., “Get Pregnant With Built On Spec Embryos Or Get Your Money Back!”
- Jessica Cussins, B.A., “Embryos for Sale: ‘When You Want Them, How You Want Them, or Your Money Back”
- Sara R. Cohen, LL.B., “It’s not about the Money: Why we are So Concerned about a California IVF Clinic’s Anonymous Embryo Program”
- Mikki Morrissette, “Creating Embryos To Sell“
My thanks to all of the authors for taking the time to discuss the issue in an open forum.
Proposed remedies to the current dilemma
From the beginning, I have been offering remedies to the embryo bank dilemma. Although far be it from me to tell CC how to run its business, these are a few ideas I had to offer:
Only patients should own embryos-
No organization, corporation or physician practice should own embryos except in the most extreme circumstances, such as embryo abandonment. With embryo donation, it is most appropriate that the donor facility simply holds the embryos, with the donors still being able to request the return of their embryos, up to the point of transfer into the recipients, should a catastrophic occurrence take place, Attorneys refer to this as being a guardian, a conservator, or a temporary holder of goods. When presenting at the American Bar Association Family Law Section Spring conference in April of 2012, many of the attorneys there strongly supported the concept of conservatorship of the donated embryos over facility ownership.
If the embryos are returned to the donor, it seems appropriate to ask the donors to reimburse the embryo donation facility for all reasonable fees expended in originally obtaining the donated embryos and returning them to the donors. We have been running our embryo donation program this way for over 12 years and we encourage others to do the same.
Excess cryopreserved embryos could be owned by patients-
As best as I can surmise for CC, their business model is to recruit a number of embryo recipients and then transfer 1-2 donor/donor embryos into each recipient. I suggest that any remaining embryos be owned by one or more of the recipients and the entire cycle should not move forward until at least one patient agrees to take the extra cryopreserved embryos, should any exist. Extra charges could be levied to those that secure the remaining embryos. In this way, no embryos remain to create an embryo bank and the CC business model remains essentially intact.
Renaming the process-
The combination of donor sperm with donor eggs and then calling them donated embryos does not fit with the ASRM definition of embryo donation (Ethics Committee of the ASRM, 2009). Embryo creation is a far better term or “shared or split donor/donor cycle” is perhaps even more appropriate. Calling such embryos donated embryos debases the amazing gift that embryo donors provide when donating their embryos.
Who should set the standards?
 SART has reviewed the concerns stated in my previous blog but I don’t think it yet has arrived at a
SART has reviewed the concerns stated in my previous blog but I don’t think it yet has arrived at a
conclusion. My understanding is that the ASRM Ethics Committee is to take up the topic during the early months of 2013. As our main guiding societies, I believe they need to take the lead, develop position statements and provide ethical standard of care guidelines for all practices to use.
Once ASRM and SART have provided ethical standard of care guidelines, I will next request that the

Canadian Fertility and Andrology Society and the European Society for Human Reproduction and Embryology (I am a member of both) consider the topic and respond with their own recommendations if they see fit.
It is not out of the realm of possibility that numerous societies could collaborate to form a consensus, such as they did when they banned the support and publication of human reproductive cloning research.
Summary comments
So where are we now on this dilemma? SART has discussed the topic but summary statements are pending. The ASRM Ethics Committee will soon meet, with the embryo bank topic apparently on the agenda. Assuming the Ethics Committee feels the topic has merit, I am uncertain how long it will take for them to release a position statement. I am hopeful that “the powers that be” will be attentive in finding a compromise that will allow CC to continue to offer their skilled reproductive services while preventing the formation of an embryo bank, no matter the size, further clarifying who should own embryos as well as the definition of embryo donation as it pertains to the current situation.
I don’t know about you but I don’t really like the idea of “McEmbryos,” or the commodification and “Wal-Martization” of human embryos. Patients should own them and decide their destiny. I am hopeful that our guiding societies will do just that – guide us on this sensitive and important topic.
Special thanks:
Thanks to Grace Centola, Ph.D., for helping to find the New York State statutes pertaining to embryo banking.
Thank you to Jessica Cussins for her blog on the topic, the reference by Professor Obasogie and her followup on the now closed Abraham Center for Life.
References:
Ethics Committee of the American Society for Reproductive Medicine. American Society for Reproductive Medicine: defining embryo donation. Fertil Steril. 2009 Dec;92(6):1818-9
.
What Impacts Embryo Donation Success Rates?

Craig R. Sweet, M.D.
Reproductive Endocrinologist

Corey Burke, B.S., C.L.S.
Laboratory Supervisor
Introduction
Asking what determines the success rates in embryo donation is an excellent question. The answer, as one might expect, is neither simple nor completely understood.
Embryologists and physicians try to choose the fewest number of healthy embryos for fresh transfers to increase success rates while minimizing multiple pregnancy rates. It is indeed a delicate balance. For example, it is well understood that in Europe, physicians transfer fewer embryos but patients also suffer significantly lower success rates than in North America. (Boostanfar R, et al. Fertil Steril 2012)
What variables do we examine to estimate probable success?
Since donated embryos are cryopreserved, the variables become even more complex compared to fresh embryos. In combining a great deal of published data and over 20 years of IVF and embryo transfer experience, we came up with what we feel are the variables which seem to influence success rates:
| Very Important! |
Preferred |
Less Optimal |
| Did the fresh cycle in which the embryos were frozen result in a pregnancy & delivery? |
Successful pregnancy and delivery |
Fresh transfer resulted in miscarriage or no pregnancy |
| Number of embryos available in a given donated set |
Four or more |
Three or fewer |
| Past implantation rates of both fresh and frozen embryo transfers |
High implantation rates |
Low implantation rates |
| Quality of the embryos frozen (link) |
High quality (Blastocyst) |
Medium quality |
| Age of the women when the eggs were provided to create embryos |
Less than 35 years old |
35 years of age or older |
| Overall health of the embryo recipient (link) |
Healthy |
With treated or untreated medical issues |
| Important |
Preferred |
Less Optimal |
| Stage of growth when the embryos were frozen (link) |
Day 5, blastocyst stage |
Day 3, 8-cell stage |
| Technique used to freeze/thaw or vitrify/warm the embryos (link) |
Vitrification |
Slow freeze |
| Overall frozen embryo transfer pregnancy rates for facility freezing the embryos |
30% or more |
Less than 30% |
| Overall frozen embryo transfer pregnancy rates for facility thawing the embryos (EDI) |
High at 30% or more |
Less than 30% |
| Ejaculated vs. surgically aspirated sperm used for fertilization |
Ejaculated sperm |
Surgically aspiration sperm |
| Somewhat Important |
Preferred |
Less Optimal |
| Was preimplantation genetic testing of the embryos done? |
Yes |
No |
| Age of the male producing the sperm |
Less than 40 years old |
More than 40 years old |
| Past successful deliveries with other embryos from donating facility |
Yes, with past deliveries |
Miscarriage, reduced survival of embryos or failed implantation |
| Probably Unimportant |
Preferred? |
Less Optimal? |
| Cryopreservation duration |
Less than 10 years? |
More than 10 years? |
The above variables influence EDI’s decision to both accept embryos from other facilities as well as determine how many donated embryos should be thawed/warmed to achieve a successful delivery.
The importance of the embryo recipient’s health should not be underestimated
In a previous blog, we described how important it was for our embryo recipients to be healthy. (link) Inadequately treated health problems, harmful medications, recreational drug use as well as smoking and weight concerns all play a potential role affecting success rates. It is clear that success depends on both the quality of the donated embryos and the overall health of the recipient.
Are all donating IVF facilities the same?
We understand that not all IVF facilities have the same success rates. Some facilities will have provided EDI  donated embryos with consistently high implantation rates while others may provide embryos of consistently lessor quality. EDI examines a facility’s past fresh and frozen embryo transfer pregnancy rates as well as its past history of providing EDI with donated embryos. It does, however, take a fair amount of time to seemingly identify a trend but we endeavor to examine all the variables we can. Our contact management database system was recently upgraded to track these variables more consistently.
donated embryos with consistently high implantation rates while others may provide embryos of consistently lessor quality. EDI examines a facility’s past fresh and frozen embryo transfer pregnancy rates as well as its past history of providing EDI with donated embryos. It does, however, take a fair amount of time to seemingly identify a trend but we endeavor to examine all the variables we can. Our contact management database system was recently upgraded to track these variables more consistently.
Do your embryos make the grade?
Understanding that all of the above variables are quite complex, we endeavored to find a simple way to convert the data into something patients could more easily understand. Since nearly all patients understand the basic A, B & C grades we used to receive in school, we modeled our grading of the embryos around these letter grades.
We created a mathematical model to assess a number of the above variables, converting the final analysis to A+, A, A-, B+, B and B- letter grades. Interestingly, we found the model really did help to predict delivery rates and continue to use it to this day to grade individual embryos as well as entire sets of donated embryos.
What information do we gain on the day of thaw and embryo transfer?
Most frequently, the embryos are thawed just hours before transfer. At times, we may thaw them days prior if, for example, they were frozen early in their development and we want to grow them further before deciding how many to transfer. Therefore, the following last set of variables will also influence success rates:
| Important |
Preferred |
Less Optimal |
| Survival rates of thawed embryos |
100% |
Less than 100% |
| Overall appearance of the thawed embryos |
Healthy, expanding and growing |
Evidence of cellular damage |
If 100% of the embryos, perhaps three out of three, survive the thaw and look healthy, we feel this is a good sign. If only 50% survive, for example perhaps only two of four, then we are concerned that the overall implantation rates will be reduced and that we might need to find more embryos to transfer just to get to the “finish line” of pregnancy and delivery. Ultimately, we want at least two high-quality embryos or up to four less certain quality embryos placed on the day of transfer.
What are the national success frozen embryo transfer delivery rates?
 In 2009, the CDC reported that there were 26,069 frozen embryo transfers performed in the U.S. with an average delivery rate of 31% per embryo transfer procedure. In addition, there were 6,074 frozen embryo transfers using embryos created from donated eggs (i.e., younger women) with slightly higher delivery rates of 34%. Please recall that donated embryos generally come from the very same types of patients listed in these success rates.
In 2009, the CDC reported that there were 26,069 frozen embryo transfers performed in the U.S. with an average delivery rate of 31% per embryo transfer procedure. In addition, there were 6,074 frozen embryo transfers using embryos created from donated eggs (i.e., younger women) with slightly higher delivery rates of 34%. Please recall that donated embryos generally come from the very same types of patients listed in these success rates.
What are EDI’s success rates?
We do our best to screen the embryos, carefully trying to choose the embryos most likely to implant. We estimate delivery rates with donated embryos to range from 27 – 42%, depending on the many variables listed in this blog, with a multiple pregnancy rates of 20-25%. We wish the success rates were higher, but please understand that frozen/thawed embryos implant and grow less frequently than fresh embryos and these percentages are entirely consistent with the frozen embryo transfer success rates described in 2009, which ranged from 31-34%.
Even with the slightly lower delivery rates compared to fresh embryo transfers, embryo donation remains one of the best and most cost-effective options for patients who cannot otherwise afford egg donation or qualify for adoption. Embryo donation still allows a woman to experience pregnancy and delivery while bonding, nurturing and protecting the ongoing gestation.
Summary
There are many variables that go into determining the potential success rates for a given set of donated embryos. First, we attempt to examine these variables carefully in deciding if EDI will accept the donated embryos. Second, we use this same information to determine how many embryos we should thaw/warm and eventually transfer. The process remains a bit of an ART (pun intended) since the complete understanding of how all of these variables influence each other and the ultimate success rates are yet to be fully known.
References
“Annual ART Success Rates.” Centers for Disease Control and Prevention. Division of Reproductive Health, 19 Apr. 2012. Web. 24 Apr. 2012. <http://www.cdc.gov/art/ARTReports.htm>.
Boostanfar R, Mannaerts B, Pang S, Fernandez-Sanchez M, Witjes H, Devroey P; Engage Investigators. A comparison of live birth rates and cumulative ongoing pregnancy rates between Europe and North America after ovarian stimulation with corifollitropin alfa or recombinant follicle-stimulating hormone. Fertil Steril. 2012 Mar 27.
How Does EDI Decide to Exclude Potential Embryo Recipients?

By: Craig R. Sweet, M.D.
Reproductive Endocrinologist
Info@EmbryoDonation.com
Introduction
There are times when we need to exclude patients when they contact Embryo Donation International (EDI) to become an embryo recipient. While not an easy decision or discussion, I thought it was time to explain our rationale when needing to, at least temporarily, exclude potential embryo recipients.
In a way, the blog we recently wrote on the ranking of potential embryo recipients dovetails into this discussion. In this blog, we described what we thought was an appropriate ranking system prioritizing those patients with the greatest need.
Our discussion here focuses on the patients who apply but who are never ranked because EDI feels they should be excluded because of any number of the reasons described below.
Excluding Potential Embryo Recipients Due to Maternal Risks
The decision to exclude a patient from embryo donation is really, in some ways, no different than the decision we have to make with other infertility patients.
Relative contraindications to pregnancy
There are occasions when certain conditions should probably be corrected before pregnancy takes place since pregnancy will often worsen or complicate the condition. Examples may include gallbladder disease and ovarian cysts or surface uterine fibroids that are five+ centimeters in average diameter. Treating these problems once pregnancy is established is very difficult, so it may be best to control the situation while we still can and correct the potential problem first before conception.
Strong contraindications to pregnancy
Patients who would be at significant risk of illness or even death should pregnancy occur include those suffering from cancer, poorly controlled systemic lupus, pulmonary hypertension or diabetes, to name a few important disease states.
Infrequently we have to be a bit paternalistic and say “no,” understanding that we may cause great harm to our ill patients by assisting them to become pregnant.
Excluding Potential Embryo Recipients Due to Risks to the Embryo/Fetus
The trickier decisions involve those patients where the potential for delivery of a live child is measurably reduced. Patients with decreased embryo implantation rates and those who are at an increased risk for spontaneous loss or at an increased risk for premature delivery/stillbirths fall into these categories.
Some of these examples are listed below:
|
Situation |
Decreased implantation |
Increased risk of spontaneous loss |
Increased risk of significant prematurity or stillbirth |
| Uterine cavity distorting fibroids or polyps |
x |
x | |
| Uterine fibroids 2+ cm in size located within the uterine muscle |
x |
x |
|
| Damaged uterine cavity with a thin endometrial lining |
x |
x |
|
| Hydrosalpinx where tubal fluid may flow back into the uterus |
x |
x |
|
| Unexplained recurrent pregnancy loss |
x |
||
| Uncontrolled medical conditions (e.g., hypertension, diabetes, renal disease, autoimmune disease) |
x |
x |
|
| History of an incompetent cervix |
x |
x |
|
| Persistent history of premature births |
x |
||
| Untreated pre-diabetes |
x |
x |
x |
| Obesity or morbid obesity* |
x* |
x* |
x |
* The effects of obesity or morbid obesity with regards to implantation rates and spontaneous loss rates are controversial.
The “Grey” Exclusion Zones
Out of the list above, one of the most difficult categories involves those patients who are obese or morbidly obese and their  potential reduction in implantation rates and increased spontaneous loss rates. Some articles show a significant reduction in implantation rates and an increased risk of spontaneous loss while others show contradictory results. While a comprehensive review of this topic goes beyond the scope of this blog, I believe there are a few things we understand:
potential reduction in implantation rates and increased spontaneous loss rates. Some articles show a significant reduction in implantation rates and an increased risk of spontaneous loss while others show contradictory results. While a comprehensive review of this topic goes beyond the scope of this blog, I believe there are a few things we understand:
- Patients with glucose intolerance and insulin resistance, regardless of weight, are at a higher risk of developing gestational diabetes during pregnancy and the potential complications associated with this disease.
- Patients who are obese or morbidly obese are clearly at risk during pregnancy for a host of issues, including preeclampsia, hypertension, large for gestational age babies, prematurity, stillbirths, gestational diabetes, Cesarean section deliveries and the risks associated with these surgeries.
There is only one preliminary study specific to embryo donation that did not show a consistent decrease in pregnancy rates as weight increased (Body Mass Index: BMI) but the trends were present suggesting an average reduction in overall pregnancy rate of 33% for obese and morbidly obese patients with a BMI of 30 or more. (Finger R., et al. 2011) We await the detailed publication of this important study to better understand this issue.
Weight loss is terribly difficult for patients and takes a great deal of time. Sometimes surgery, such as a gastric band or intestinal bypass surgery, may be the best option. These issues are the thorniest to decide, with pressure applied by potential embryo recipients who do not fully understand how their weight may contribute to failed implantation or pregnancy loss, though it certainly places them and their unborn offspring at greater risks during the pregnancy.
Surrogacy as an Option When Material/Embryonic/Fetal Risks are Too High
 Some of the issues discussed in this blog can be treated. Then the potential embryo recipient can be moved out of the exclusion zone. Some of these issues are not treatable, so options such as surrogacy and/or adoption may be better alternatives.
Some of the issues discussed in this blog can be treated. Then the potential embryo recipient can be moved out of the exclusion zone. Some of these issues are not treatable, so options such as surrogacy and/or adoption may be better alternatives.
While some embryo donation programs refuse to allow surrogacy, EDI feels this is an excellent alternative. For example, is it ethical for EDI to ask that the patient with asymptomatic uterine fibroids, which may significantly reduce implantation or increase pregnancy loss rates, be surgically removed in a patient who fears surgery? Is gestational surrogacy a better alternative?
In Summary
We do not mean to be cruel or judgmental but are forced to make difficult decisions regarding the acceptance or exclusion of embryo recipients. We owe it to the potential embryo recipients to give them the best chance possible, understanding there are medical conditions that may severely impair their chances for success. We owe a debt of gratitude to the embryo donors and take seriously the responsibility of finding a healthy patient for their embryos hoping to maximize the chances that the embryos will survive and thrive. Lastly, and certainly not least, we owe it to the embryos to make certain they have the best chance possible.
Unfortunately, we sometimes have to make the difficult decision to exclude a potential embryo recipient, at least temporarily, until the medial concerns are remedied or certainly improved.
References:
Finger R, et. al. Obesity and the ability to achieve pregnancy in embryo donation. Fertil Steril 2011;96(3)-S172.
Parent via Egg Donation – Parent via Embryo Donation

Marna Gatlin of PVED
Exploring what we know – Marna Gatlin, Founder Parents via Egg Donation and guest blogger
I was asked recently what I thought about embryo donation vs. egg donation. Is a parent via egg donation the same as a parent via embryo donation? What do they have in common? Or are they very different?
My knee jerk reaction was “Well doh, of course they are the same. They are both embarking upon a unique journey to become parents right?”
When I thought more about it, the word “versus” jumped out at me. Did the individual asking the question really mean “versus” as in against, or in contrast to?
Are the two mutually exclusive?
When I think of egg donation and embryo donation, I think about the word “AND” – I don’t think of it as an either or. Both are just different ways of either growing or adding to your family.
Some approach embryo donation with great trepidation because they worry about the bonding process, or about the explanation or story they will be sharing with their child. The reality is – the path might be different but at the end of the day the goal is the same – you are becoming a parent.
Let’s delve a little deeper. If you receive a donor egg, the genes of your baby are going to be combined with the genes of your husband (or partner) and those of your egg donor. Or if you are a single mother, or women in a same sex relationship, the donor egg will be combined with donor sperm. You will probably undergo what we call a fresh embryo transfer. You will carry that baby for nine months and then deliver that baby. Women often wonder if there’s a down side to carrying a baby that is not genetically related to them. You know this is an age old question that’s been asked and answered since the early 80’s when the first donor egg child was conceived, carried, and delivered. I can tell you as a parent via egg donation myself that it doesn’t matter. I carried my son for nine months. The bond I created with him is rock solid, loving, and he’s my son. The lack of genetic connection simply doesn’t matter. Not one iota. The very same thing happens with embryo donation recipient parents who are receiving truly a meaningful gift of life.
genes of your husband (or partner) and those of your egg donor. Or if you are a single mother, or women in a same sex relationship, the donor egg will be combined with donor sperm. You will probably undergo what we call a fresh embryo transfer. You will carry that baby for nine months and then deliver that baby. Women often wonder if there’s a down side to carrying a baby that is not genetically related to them. You know this is an age old question that’s been asked and answered since the early 80’s when the first donor egg child was conceived, carried, and delivered. I can tell you as a parent via egg donation myself that it doesn’t matter. I carried my son for nine months. The bond I created with him is rock solid, loving, and he’s my son. The lack of genetic connection simply doesn’t matter. Not one iota. The very same thing happens with embryo donation recipient parents who are receiving truly a meaningful gift of life.
The most beautiful aspect of embryo donation to me is that embryos that are being placed for donation are done so purposely. These are embryos that the donating parents know have created amazing children who are loved, honored and cared for. These donating parents want to make sure their embryos are donated to a home that will love, cherish and honor the resulting children as they would. Regardless of whether an individual has a child via egg donation or embryo donation, the fears of parenthood almost always focus on the unknowns. And here’s a secret: they are also experienced by those who are conceived naturally. Moms and Dads all over the world have the same worries about parenting as do parents via egg donation and embryo donation.
Parents via egg donation often ask questions such as:
- Am I going to screw up my child?
- Will my child love me?
- How am I going to relate to my child?
- Will my parents and other family members accept my child?
- How and when will I share information about their conception?
When we look at embryo donation the questions that we find unique to embryo donation are:
- Are we protected legally, can the donating parents come back and claim our child in the future? (The answer is NO, they cannot. That’s why it’s important to have a clear legal contract.)
- Will my child have access to information about his or her health in the future?
- Will my child have siblings and if so, will they have the opportunity to know them?
- Will my child or children see me as their real parent?
- How will I explain this to my family or friends?
- What about stupid comments from those around me?
- How and when will I share the information about my child’s conception with my child?
To me there is no difference between being a parent via egg donation or a parent via embryo donation. The end result is the same.
At the finish line we are simply Mom and Dad.
Marna Gatlin
Parents via Egg Donation
www.PVED.org | marna(at)pved.org
A little bit about Marna…
After many years of struggling with infertility, PVED founder Marna Gatlin discovered that the technology to have a child through egg donation was available. She was curious, excited, and above all, hopeful that this process might be the conduit to finally achieving her lifelong dream of becoming a parent.
Marna ensures that all the needs of egg donor recipients are met, maintaining a high standard of ethics and confidentiality. Marna advocates and assists recipient parents, helping them arrange for the highest quality patient care, wherever in the world they reside. Her experience and knowledge related to the complex emotional and physical needs of individuals dealing with infertility makes her an essential asset PVED.
As a previous recipient, Marna is uniquely qualified to provide caring and timely services. Marna is truly dedicated to compassionately guiding couples experiencing infertility through their treatment process.
Marna is joined by several dedicated and knowledgeable support staff that all work together clearly dedicated to see the success of PVED. These include clinical psychologists, reproductive endocrinologists, attorneys, as well as a talented business and public relations team.
Marna attended Eastern Oregon University and Portland State University majoring in Business, Psychology, Social Science, and is a member of the American Society of Reproductive Medicine (ASRM), the European Society of Human Reproduction and Embryology (ESHRE) and the Society for Assisted Reproductive Technology (SART). Marna, a writer, is married, has a son, and does some of her best thinking and creating atop of her John Deere tractor mowing and cultivating her back forty.
Do These Donated Embryos Make the Grade?
Embryo Grading Made Easy For Embryo Donors and Embryo Recipients
Part 1 of a 3 part mini-series by Corey Burke, B.S., C.L.S. & Laboratory Supervisor and Reproductive Endocrinologist Craig R. Sweet, M.D.
Embryo grading is an important factor for both donors and recipients. Potential embryo donors want to know if we will accept their embryos for donation since part of our decision is based on the grade of their embryos. Likewise, potential embryo recipients want to know how likely the donated embryos are to survive thawing and if they are of good enough quality to build their family. Part of our estimation of success depends on the grade of the embryos.
We wish this process could be easier since there is no standardized system used in all embryology laboratories to grade embryos. Furthermore, the grading of embryos is somewhat subjective so one embryologist may grade an embryo
somewhat differently than a colleague.
Embryo grading is an imperfect process; poorly graded embryos may occasionally result in ongoing pregnancies and beautiful looking embryos may not implant and grow. Poorer graded embryos will not necessarily result in an abnormal child; they simply seem to implant and grow less frequently.
So, the appearance and grading of an embryo is an imperfect estimate of the quality of the embryo as well as the embryo’s true potential. It is, however, the best way we have to visually estimate the implantation and live birth rate of a given embryo. We will now examine one of the more common methods used to grade embryos.
When are embryos graded and cryopreserved?
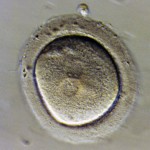
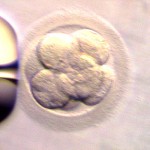
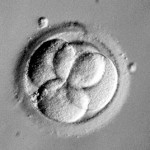
Embryos are usually graded and frozen at three specific stages with “Day 0” being the day of retrieval and fertilization:
| Age of Embryos | Day 1 | Day 3 | Day 5-6 |
| Common terminology | 2PN (pronuclear stage)
2 cell stage |
Embryos are 6-10 cells | Morula &
Blastocysts |
| Grading importance | Grading not available | Grading relatively important | Grading very important |
| How often these are sent to EDI? | Rarely sent | 40% of EDI embryos | 60% of embryos |
Embryos cryopreserved immediately after fertilization are confirmed early on Day 1 (the 2PN (pronuclear stage), but aren’t advanced enough to be consistently graded. Freezing embryos on Day 1 is quite infrequent unless we are certain there will not be an embryo transfer. Accordingly, these embryos are rarely sent to EDI for embryo donation.
Grading Day 3 Embryos
Day 3 embryos are graded on cell number, the amount of cellular fragmentation and the symmetry of the embryo.
Cell Number
Most Day 3 embryos will be comprised of 6-10 cells called blastomeres. Embryos with too few blastomeres may not be healthy, so we prefer at least 7-8 at this stage. Embryos with fewer cells may not be healthy growing very slowly or may have stopped growing entirely commonly called “cell block”.
Fragmentation
Fragments may be found in many of the embryos, which are “bits” of cells that break off from a blastomere. We prefer as little fragmentation as possible. Fragmentation is estimated as the percentage of fragmentation volume compared to the total embryo volume and is converted to a letter grade in the following way:
- 0 % = A
- 1-10% = B
- 11-25% = C
- >25% = D
A large amount of fragmentation may be caused by death of one or more of the blastomeres. The higher the fragmentation, the lower the quality of the embryo & letter grade and the less likely that the embryos will survive thawing. Embryos with high fragmentation rates implant less frequently when transferred fresh or when thawed.
Symmetry
Symmetry of the embryo refers to the shape of the individual blastomeres and the overall shape of the embryo. The blastomeres should all be very similar in size and generally round in shape. The scale used to grade symmetry is
- Perfect = A
- Moderately asymmetric = B
- Severely asymmetric = C
Severe blastomere asymmetry (i.e., large and small blastomeres in the same embryo) reflect nuclear/chromosomal and cytoplasmic problems suggesting the embryo is less healthy than desired. There is supporting evidence that blastomere symmetry is important and reflects overall health of the embryo. Interestingly, the overall symmetry of the embryo (round vs. oval) is of uncertain importance with some very “funny looking” embryos resulting in beautiful and healthy children.
Putting it All Together for Day 3 Embryos
For Day 3 embryos, the order of grading is the “number of cells (#c),” “fragmentation grade” and “symmetry grade”. For example:
- 7cAA = 7 cells with no significant fragmentation and perfect symmetry
- 8cBA = 8 cells with 1-10% fragmentation and perfect symmetry
- 6cBB = 6 cells with 1-10% fragmentation and moderate asymmetry.
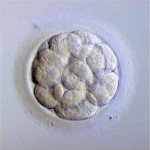
Grading Day 5 Embryos
More advanced embryos are graded and potentially frozen on Day 5 or Day 6. These are generally described as morula or blastocysts.
Day 5 Morula Embryos
Morula embryos are difficult to grade as the cells combine, forming essentially a ball of cells that can’t really be categorized in any way other than descriptive terms:
- Morula (early)
- Compacting morula (more advanced)
While some facilities only occasionally cryopreserve Day 5 morula embryos, it is thought that the survival and implantation rates of these embryos may be slightly reduced but they are still quite reasonable, suggesting that they should not be discarded. Day 6 morulas are probably delayed in growth or may have stopped growing, may not be viable and are infrequently cryopreserved.
In order to balance the possible reduced implantation rates, it is common that more morula embryos are thawed and transferred in order to achieve success.
Day 5 Blastocyst Embryos
Day 5 blastocyst embryos are the most advanced embryos we see in IVF. These embryos are formed within 24 hour of actual implantation. Trying to grow embryos beyond this point is
technically difficult, as the embryos usually don’t survive. In addition, the window of time for implantation seemingly closes beyond Day 5 or early day 6. For example, transfer on Day 7 will rarely result in implantation. So, it simply makes more sense to transfer and/or freeze blastocysts rather than trying to grow them any further.
Along with descriptive measures, more objective grading is attempted through evaluation of the cellular expansion, the inner cell mass (which eventually becomes the fetus) and the quality of the outer cell mass called the trophectoderm (which eventually forms the membranes and placenta).
Expansion
As the embryo advances in growth, a cavity called the blastocoel fills with fluid. As the cells continue to divide and the fluid collects, the embryo expands and eventually escapes its outer covering called the zona pellucida. The blastomeres continue to group together wherein the individual cell cannot be counted. As the Day 5 embryo expands, differentiates and escapes the outer zona pellucida, the grade increases numerically from 1-5.
| Grade | Description | Physiology |
| 1 | Early Blastocyst | Starting to form a fluid-filled space in the middle (Blastocoel). Grading the embryo is difficult here. |
| 2 | Full Blastocyst | Blastocoel forms and inner cell mass is now distinguishable. Grading can be done from this point forward. |
| 3 | Expanded Blastocyst | Blastocyst is starting to expand in size thinning the outer covering, the zona pellucida |
| 4 | Hatching Blastocyst | Blastocyst is starting to hatch out of the zona pellucida. |
| 5 | Hatched Blastocyst | Blastocyst is fully hatched and now ready for implantation into the uterine wall. |
Inner Cell Mass (ICM)
As the blastomeres compact to form the inner cell mass (ICM), this early fetal tissue is graded on a A-D scale:
| ICM Grade | Description |
| A | ICM with total compaction |
| B | ICM still compacting |
| C | Reduced ICM |
| D | Poor with dying cells |
Trophectoderm (TE)
The outer cells of the trophectoderm (TE) also reflect the overall health of the embryo and are graded in an A-D scale.
| TE Grade | Description |
| A | Numerous cells forming cohesive layer |
| B | Few but healthy large cells forming a loose epithelium |
| C | Few cells present often with asymmetric distribution |
| D | Poor with degenerating/dying cells |
Putting it All Together for Day 5 Embryos
For Day 5 embryos, the order of grading is “expansion,” “inner cell mass” and “trophectoderm.” For example:
- 1 = Early blastocyst is unable to be easily graded with respect to ICM or TE as these haven’t separated well enough yet.
- 2BB = Blastocyst with partial ICM compaction with loose, large trophectoderm cells
- 3AB = Expanding blastocyst with total compaction of ICM and with loose, large trophectoderm cells
- 4AA = Hatching blastocyst with excellent ICM & trophectoderm cell layers
- 5AA = Fully hatched blastocyst with excellent ICM & trophectoderm cell layers
Day 5 embryos that are graded 4AA and 5AA are some of our favorite embryos.
Summary Comments
Embryology laboratories strive to grow the healthiest embryos they can. Over time, they have adapted different grading techniques so the laboratories can communicate the quality of the embryos to physicians, patients and each other. Not all beautiful embryos will implant or produce a healthy child but they seem to do so more often than others. Not all poorly developing embryos will fail to implant and produce a healthy child but most do not result in live offspring.
At EDI, we try to only accept embryos that are likely to implant so embryos with less than a B rating for any category are infrequently accepted. This is done to assure our recipients that all of the donated embryos we offer are of the highest quality and provided the greatest chance for a successful pregnancy.
This is only one piece of the puzzle as many other factors influence the likelihood for success. Dr. Sweet will cover this topic in the separate blog within the next couple of months.
Also stay tuned to the upcoming blogs regarding how embryos are frozen and thawed and what techniques seem to work the best.
While embryo grading is not a perfect system, we use it to try to predict the overall quality of the embryos and their potential to survive thaw, grow and build a recipient’s family.
Corey Burke. B.S., C.L.S.
Laboratory Supervisor
CBurke@EmbryoDonation.com
Craig R. Sweet, M.D.
Reproductive Endocrinologist
Info@EmbryoDonation.com
References
Racowsky C, Vernon M, Mayer J, Ball GD, Behr B, Pomeroy KO, Wininger D, Gibbons W, Conaghan J, Stern JE. Standardization of grading embryo morphology. Fertil Steril. 2010 Aug;94(3):1152-3.
New Mini-Series on How Embryos are Processed
 Beginning next week, we’ll be publishing a mini-series on how embryos are processed in the laboratory. Written by Corey Burke, B.S., C.L.S. & Laboratory Supervisor and Reproductive Endocrinologist Craig R. Sweet, M.D., the three-part series will cover how embryos are initially graded, frozen and finally thawed. The information is designed for patients and written in an easy to understand style.
Beginning next week, we’ll be publishing a mini-series on how embryos are processed in the laboratory. Written by Corey Burke, B.S., C.L.S. & Laboratory Supervisor and Reproductive Endocrinologist Craig R. Sweet, M.D., the three-part series will cover how embryos are initially graded, frozen and finally thawed. The information is designed for patients and written in an easy to understand style.
We hope this will be an informative series, answering many common questions alike. If you have additional questions, please feel free to post them as comments here, or after the specific blog post.
Thank you for reading and stay tuned for detailed embryo grading, freezing and thawing information starting with the first series post on Tuesday, January 10th.
–Embryo Donation International
How Does Embryo Donation International Rank Embryo Recipients?
Unfortunately, there are far more potential embryo recipients than donated embryos. While in this country alone, hundred of thousands of embryos are stored in liquid nitrogen, (Hoffman DI, et. al, 2003), less than ten percent and frequently less than five percent are donated to patients in need (Klock SC, et al., 2003). As a result, we often have a waiting list of recipients asking for donated embryos.
First, EDI doesn’t discriminate with respect to race, religion, ancestry, sexual preference or  marital status. We also try to be fair to existing EDI patients and to those who have yet to become established patients. We basically are looking for potential embryo recipients who have few other reproductive options available. If one views donated embryos as an essentially rationed commodity, we want to make certain that patients in greatest need are ranked the highest. We try to do this in an ethical and fair fashion but ranking them can be a challenge.
marital status. We also try to be fair to existing EDI patients and to those who have yet to become established patients. We basically are looking for potential embryo recipients who have few other reproductive options available. If one views donated embryos as an essentially rationed commodity, we want to make certain that patients in greatest need are ranked the highest. We try to do this in an ethical and fair fashion but ranking them can be a challenge.
While not absolute, below is a general ranking from high priority to lower priority in our general waiting lists:
- The intended parents have no delivered children and limited financial means with few options available.
- Their only child died and no other siblings exist.
- One of the partners raised a child with a different partner.
- Both of the partners raised children with different partners.
- Both partners raised one adopted child together.
- Both partners raised one (genetic) child together.
- Both partners raised children with other partners and one (genetic) child together.
- Both partners raised multiple (genetic) children together.
We define a “genetic child” as a child whose genes came from the parents themselves without the use of donated material and also not an adopted child.
Here are some of the questions we also commonly ask in trying to decide where the patients should rank on our waiting lists:
- How long have they been on the waiting list?
- Are they established patients with EDI or have never been seen before?
- Are there other less expensive and viable options such as donor sperm?
- What have the patients gone through during their infertility journey?
- Is adoption of a live child a possibility?
- Are there financial constraints that makes embryo donation far more feasible over other more expensive options?
Also, we actually have multiple lists broken down by the following major categories:
- Type of embryo donation: Anonymous, Approved and Open Embryo Donation
- Marital status/sexual orientation: Single woman, single man (very rare), heterosexual couple (married/unmarried), lesbian couple and gay couple (married/unmarried).
 As you can see, the process of ranking can get a bit complicated. Our highest priority is healthy embryo recipients who have never had children of their own and have very few fertility options with limited financial means. For example, this group may include cancer survivors who have been left infertile due to the disease or treatment. In many states, cancer survivors find adoption of a child extraordinarily difficult, so embryo donation may be their only option for building a family. We also tend to rank patients higher who have undergone extensive unsuccessful IVF treatments and egg donation is their only remaining option, but due to financial constraints, are unable to afford the procedure.
As you can see, the process of ranking can get a bit complicated. Our highest priority is healthy embryo recipients who have never had children of their own and have very few fertility options with limited financial means. For example, this group may include cancer survivors who have been left infertile due to the disease or treatment. In many states, cancer survivors find adoption of a child extraordinarily difficult, so embryo donation may be their only option for building a family. We also tend to rank patients higher who have undergone extensive unsuccessful IVF treatments and egg donation is their only remaining option, but due to financial constraints, are unable to afford the procedure.
While we do not exclude patients who have raised children, some requests come from parents with numerous children in their current family. We are actually not looking for potential embryo recipients who are trying to “save” donated embryos. We wish we had enough embryos for everyone, but until we have more embryos than applicants, we will continue to prioritize patients who have never raised children higher than those who have previously experienced the joys of parenthood. We will not exclude such patients, they will be ranked far lower than others on the priority scale.
While some patients who approach EDI have no children, they may have several other fertility options. For example, a young couple that is infertile due to severe male factor infertility, may best be served with sperm donation. Donor sperm generally is less expensive and faster than embryo donation and they can have more than one child using the same donor. We would prefer to save the donated embryos for patients with few available options.
We hope that our readers will understand our basic goals. For any rationed commodity, we want to save it for those in greatest need. If one pictures the embryos as rare, precious and hard to come by, one can imagine why a fair ranking system must be developed.
Ranking systems are never perfect and often seem unfair to those who receive lower rankings. It is difficult to explain a lower ranking to a patient who is desperately seeking to build or expand their family, however, once separated from the emotional aspect, it is usually agreed that a ranking system is needed to fairly as many patients that we can who are seeking donated embryos.
In summary, we feel that a ranking system is necessary to achieve our goal of matching donated embryos to patients in need in an ethical and fair fashion. In a perfect world, infertility wouldn’t even exist and there would be enough donated embryos to meet the demands of those asking for donated embryos. In today’s realistic world, however, the precious gift of donated embryos is simply not given often enough to meet the demands of those seeking donated embryos. Perhaps someday ,though education and research, the number of patients offering their embryos for donation will increase to such a level that we may be able to eliminate or at least trim back segments of our ranking system. Until that day comes, we will have to do our best to assist in matching patients in need to donated embryos in the most ethical and fair fashion we can.
Shelley Osking, L.P.N.
Embryo Donation Coordinator
Shelley@EmbryoDonation.com
Corey Burke, B.S., C.L.S.
Laboratory Supervisor
CBurke@EmbryoDonation.com
Craig R. Sweet, M.D.
Reproductive Endocrinologist
Founder, Medical and Practice Director
Info@EmbryoDonation.com
Hoffman DI, et al. Cryopreserved embryos in the United States and their availability for research. Fertil Steril 2003;79:1063-9.
Klock SC, et al. The disposition of unused frozen embryos [letter]. N Engl J Med 2001;345(1):69-70.
Georgia Law: The First Salvo Towards Embryo Personhood
A Guest Post by: Harold Eskin, Esq.
New laws are beginning to appear on the books of many states that support “embryo adoption.” “Embryo Adoption” is placed in quotation marks because that phrase alone has unique connotations that I discuss briefly below. The term embryo “donation” is also a commonly used expression describing giving one patient’s/couple’s cryopreserved (frozen) embryo(s) to another patient/couple trying to expand their family, but who otherwise have been unsuccessful through natural and/or advanced reproductive techniques.
 Georgia was the first state to pass an embryo adoption bill (2009) that provided an opportunity for intended parents to go through an adoption procedure to obtain the right to gestate a thawed frozen embryo. Other states, such as Florida, have embryo donation statutes on its books, which allow a couple to receive a donation from another (open or anonymous) of a frozen embryo. While the end results may appear to be the same, the road getting there and implications of using the different phrases are vastly different.
Georgia was the first state to pass an embryo adoption bill (2009) that provided an opportunity for intended parents to go through an adoption procedure to obtain the right to gestate a thawed frozen embryo. Other states, such as Florida, have embryo donation statutes on its books, which allow a couple to receive a donation from another (open or anonymous) of a frozen embryo. While the end results may appear to be the same, the road getting there and implications of using the different phrases are vastly different.
The Georgia law, which was championed by Right to Life groups, treated a frozen embryo in much the same way as it would a child already born. The new law and procedures mimic that of other adoption provisions and gives the frozen embryo many of the same rights and considerations of a born child, including using a “best interest of the child” standard in the adoption analysis. This philosophy is consistent with the concept that a child’s rights (as compared to the mother’s) begins at conception rather than birth and has implications in the abortion-right to choose/right to life arguments ongoing disagreements and potential laws expressing same.
Florida and many other states have historically treated frozen embryos as the property of the parents, who have the right to donate or dispose of the frozen embryos as they saw fit. The recipients received the frozen embryos as property under the respective laws of their state and could use or dispose of the frozen embryos as they saw fit. This process allowed for freer access to unused frozen embryos and discouraged the abandonment/discarding of them.
The agenda of the Georgia law was not necessarily meant to “protect” the frozen embryos but was designed to advance a political agenda of creating additional barriers to a women’s right to choose (i.e. restrict abortion) and to further control the reproductive rights of patients by discouraging the use of advanced reproductive techniques, such as in vitro fertilization as well as the cryopreservation and storage of excess embryos.
Up to now, there have been few attempts to export the Georgia concept in other states. This is perhaps due to the country’s economic challenges, but this possible trend needs to be closely monitored. The implications of providing “personhood” to embryos are far and wide and the Georgia statute is one of the first successful salvos to be launched with others most certainly to follow. Mississippi is currently targeted for a constitutional amendment to give embryos personhood and many other states are next in line for challenges that may significantly impair the health and reproductive care of women.
Harold Eskin, Esq.
www.LegalSurrogacy.com
HalEskin@LegalSurrogacy.com
1420 SW 47th Street
Cape Coral, FL 33904
239-549-5551
References:
Georgia Statute:
http://statutes.laws.com/georgia/title-19/chapter-8/article-2/19-8-41Florida Statute:
http://www.leg.state.fl.us/Statutes/index.cfm?App_mode=Display_Statute&Search_String=&URL=0700-0799/0742/Sections/0742.14.html
Hope & Will Have a Baby – Book review by Craig R. Sweet, M.D.
 This book is one of a collection of third party conception children’s books written by the same author and illustrator and published by Graphite Press, copyright of 2006. It is one of the only books I have seen for children conceived though embryo donation.
This book is one of a collection of third party conception children’s books written by the same author and illustrator and published by Graphite Press, copyright of 2006. It is one of the only books I have seen for children conceived though embryo donation.
Ms. Celcer is a mental health professional that feels it is important to be up front in telling the children of embryo donation about their origin. In her forward, she cites the past secrets in adoption causing harm. She goes on suggesting that-
- • Children somehow sense the secrets
- • The failure to divulge may create shame in the parents of donated embryo offspring
- • Failure to tell a child results in a loss of pride of their beginnings
This book is to be used to tell the children about their loving conception while trying to manage the complex emotions of the parents.
Through the eyes of Hope and Will, a young married couple in love, the book explains what a “special place” the uterus is and how  embryos are created. Hope and Will experience infertility, depression and probably some grumpiness (can you imagine?). They seek help through Dr. Quest, who gives Hope tablets, pills and shots, but to no avail. Embryo donation is discussed, explaining anonymous and open options in simple and easy to understand terms.
embryos are created. Hope and Will experience infertility, depression and probably some grumpiness (can you imagine?). They seek help through Dr. Quest, who gives Hope tablets, pills and shots, but to no avail. Embryo donation is discussed, explaining anonymous and open options in simple and easy to understand terms.
Hope and Will conceive though embryo donation. The author emphasizes love, pride and excitement when the delivery finally takes place. Hope and Will are clearly grateful to their donor couple whom they have never met. Enough information is provided about the donors to help the child understand his origin.
While perhaps a little complex for a young child of four or five, the book might work quite well for a slightly older child. The pictures are wonderful and the emphasis on love and the desire to have a child by whatever means necessary is well done. I have always suggested that if parents tell the child, they should highlight their tremendous desire to have and love that child, as well as how hard they worked to bring such a wonderful child into their home.
Disclosure in embryo donation is a periodic theme of a number of my blogs. Adoption is clearly different than embryo donation. Adoption tends to be well accepted by most of the world’s religions. If embryo recipients tell friends, family and their donor-conceived offspring, there may be significant repercussions. Will the recipients and offspring be criticized, ostracized or, even, excommunicated by these same people or by their own religion?
Unfortunately at this time, I simply don’t believe we have enough data to make a clear recommendation to embryo recipients although there is wonderful ongoing research that will hopefully help to answer this question. For now, extrapolating what we have learned from adoption may simply not be appropriate for embryo donation, although passions on both sides of this issue  abound. The long-term consequences of secrecy vs. total openness to friends, family and the child of embryo donation are essentially unknown and I feel this decision needs to be made carefully with consultation with skilled mental health professionals (and Ms. Celcer is one of them), reproductive endocrinologists and perhaps, most importantly, recipients that have already traveled this journey.
abound. The long-term consequences of secrecy vs. total openness to friends, family and the child of embryo donation are essentially unknown and I feel this decision needs to be made carefully with consultation with skilled mental health professionals (and Ms. Celcer is one of them), reproductive endocrinologists and perhaps, most importantly, recipients that have already traveled this journey.
Selling for about $20 at Barnes & Noble, the purchaser should know this is a paperback and only 28 pages. The reader is clearly paying for the content and not story length or volume.
I highly recommend this book for those parents who decide to tell their child they were conceived through embryo donation, although the story line may need to be changed for single women and gay and lesbian couples. For the lucky parent(s) of embryo donation, this is a fairly good place to start if disclosure is the path they choose to follow.
Craig R. Sweet, M.D.
Founder, Medical & Practice Director
Embryo Donation International
Disclosure Issues in Embryo Donation: Summary Comments
Brief Introduction
Embryo donors and recipients are faced with a number of life-changing decisions as they contemplate disclosure. This is the final segment of a five-part series summarizing the complex decisions surrounding the disclosure of the genetic origins of embryo donor-conceived individuals to family, friends and the offspring themselves.
What are some of the questions that embryo donors and recipients must explore before deciding to disclose or not?
Embryo donors must decide if they want a relationship with the donor-conceived individual being raised by another family. Are the donors willing to disclose their donation of embryos, which may have taken place years ago, to friends and family, including their existing children? At what age would they want the donor-conceived individual to contact them?
Recipients need to ask related questions about whether they should tell their embryo donor-conceived children about their origins. If they want to do so, when should they tell and what information should they share? Are the recipients comfortable letting their family members and friends know of their infertility history? Will the recipients and offspring be criticized, ostracized or, even, excommunicated by these same people or by their religion? Are they comfortable with their child contacting and potentially having a relationship with the embryo donors and their blood siblings?
Can an understanding of adoption disclosure be used in the world of embryo donation?
It is understandable, but potentially misguided, that some professionals believe embryo donation is identical to the adoption of a live child. I have written about this topic before. While the American Society for Reproductive Medicine (ASRM) states that the term “embryo adoption” should not be used, some facilities do so openly.
However, regardless of the legal differences, there also are a number of very practical differences:
- Because the embryo recipient carries and delivers the child, her pregnancy may look natural, making disclosure by the recipients optional. Adoption is much more difficult to hide.
- Disclosure of the embryo donation process may not be well accepted by friends, family and some religions. While not universally accepted, adoption may be better tolerated.
- As with sperm donation, single women and lesbian couples who conceive with donated embryos are more likely to disclose because the fatherhood issue inevitably arises. Heterosexual couples, however, are the least likely to disclose.
How often is disclosure currently revealed?
 There is a growing body of data that suggests only a minority of the embryo donor-conceived children are told of their origins. In an English study of 17 embryo donation families with donor offspring between five and nine years old, only 18% of the recipient parents had told their children (MacCallum F. et al. 2008). An additional 24% planned on telling, 12% were undecided and 47% stated they would not tell. The reality is that many of those who planned on telling or were undecided may eventually decide against disclosure as the child ages and enters the more difficult years of adolescence. This seemed to be different than other donor procedures where 46% of donor sperm insemination parents and 56% of egg donation parents planned to disclose (Golombok S, et al. 2004).
There is a growing body of data that suggests only a minority of the embryo donor-conceived children are told of their origins. In an English study of 17 embryo donation families with donor offspring between five and nine years old, only 18% of the recipient parents had told their children (MacCallum F. et al. 2008). An additional 24% planned on telling, 12% were undecided and 47% stated they would not tell. The reality is that many of those who planned on telling or were undecided may eventually decide against disclosure as the child ages and enters the more difficult years of adolescence. This seemed to be different than other donor procedures where 46% of donor sperm insemination parents and 56% of egg donation parents planned to disclose (Golombok S, et al. 2004).
Why has anonymity been the norm in embryo donation?
The concept of anonymity in egg/sperm/embryo donation has its roots in the physician/patient relationship to meet a series of needs (Daniels K. 1997). To assume that it should be discontinued for everyone doesn’t take into account the ethical, social and religious circumstances of different patients.
Since the medical profession assumes parents are able to make, without question, thousands of different choices for their children, assuming they are incapable of making a decision, such as disclosure which affects both the child and parents, is simplistic and rather paternalistic.
Why not disclose?
Let’s face it; there might indeed be a lack of societal approval of offspring who originated from donor material. Many people are judgmental regarding embryo donation, especially in cultures and religions that emphasize genetic inheritance. Recipients have legitimate reasons to fear the potential damage to themselves and their children from other people’s negative reactions, social stigma and resulting isolation (Shehab D, et al. 2008).
Some of the reasons recipients do not want to disclose include protecting their children  and family relationships from rejection, not feeling a need to disclose or uncertainty about how to approach the matter (MacCallum F, et al. 2007, Jadva V, et al. 2009 & Mahlstedt PP, et al. 2010). Disclosing also will broadcast their infertility issues, which they may have kept quite private in the past (Klock SC. 1997). Another common reason given for not disclosing is fear that they as non-genetic parents will be rejected. Actually, there is no data to support or deny this last very human concern.
and family relationships from rejection, not feeling a need to disclose or uncertainty about how to approach the matter (MacCallum F, et al. 2007, Jadva V, et al. 2009 & Mahlstedt PP, et al. 2010). Disclosing also will broadcast their infertility issues, which they may have kept quite private in the past (Klock SC. 1997). Another common reason given for not disclosing is fear that they as non-genetic parents will be rejected. Actually, there is no data to support or deny this last very human concern.
Unlike adoption, it is doubtful that the offspring of embryo donation will have to resolve the “history of rejection” unlike adopted children who were separated from their birth parents (Widdows H, et al. 2002). Families created through embryo donation are a product of a loving gift and not formed from perceived rejection. Therefore, some of the motivation to disclose in adoption simply does not exist in embryo donation.
Why should parents disclose?
The most common reason for embryo recipients to disclose is a fear that the child will accidently discover the facts at a later date (MacCallum F, et al. 2007). One study of adult offspring of sperm donation found that about 1/3rd of the individuals learned of their donor origins after an argument, from another person or they just figured it out themselves (Mahlstedt PP, et al. 2010). In a recent study, about 10% (47/458) of the sperm donor offspring who were searching for their donors and half-siblings found out by accident (Beeson DR, et al. 2011). They were apparently told by siblings, family or friends; discovered paperwork or inheritable medical issues that their recipient parents did not have; inadvertently overheard their parents talk about it; or it was revealed as a consequence of divorce. Recipients who tell family and friends but not the child are asking for future problems. When examining these two studies, it is clear there is a reasonable risk that the offspring will discover their origins, even under the best of circumstances. It also is important to realize that sperm, egg and embryo donation procedures are different when analyzing the study results.
There are some embryo donation recipients who feel it is simply best to not have secrets in the home. Others believe the embryo donor-conceived children have a moral right to know their origins. Some feel it is important that the children understand and connect to their genetic inheritance beyond the family who raised them.
What should be disclosed?
There are three decision levels in disclosure: 1) To decide to disclose, 2) To decide what age to disclose, and 3) To decide what information is to be provided. One can simply tell the child he or she was embryo donor-conceived but the child is certain to have more questions. At the very least, I believe providing the donors’ photographs (when they are available) as well as their medical, surgical, psychiatric, family and social histories are needed. However, providing actual identifying information needs to be handled carefully and thoughtfully.
When should disclosure take place?
Disclosure early in the child’s development, certainly before age ten, would seem to be ideal. Some mental health professionals (MHP’s) feel it should be discussed as early as possible. If disclosure occurs in adolescence or later, young adults may feel mistrust, alienation, identity confusion, frustration and even hostility towards their family (Ethics Committee, 2004 & Mahlstedt PP, et al. 2010). About 46% of donor sperm offspring who were told at the age of 18 or older stated they were confused. It is probably best to tell the child before the age of 10 than wait until later where there is a doubling of the number of children who were unsettled with the information.
Why do embryo donor-conceived individuals want to know their genetic origins?
Many embryo donor-conceived individuals may be curious about their donors’ physical characteristics and original motivation to donate, as well as possess a desire to know their genetic identity and, perhaps, provide an ancestral history for their own children. (Ravitsky V, et al. 2010 & Mahlstedt PP, et. al. 2010).
For many, the search for their donors may go well beyond just seeking simple information. In two recent studies of offspring searching for their donor sperm fathers, 80-88% were intensely curious about their donor and wanted to contact him. Up to one-third desired an actual relationship with the sperm donor (Beeson DR. 2011 & Mahlstedt PP, et al. 2010). It should be understood, however, that studies such as these might not represent a balanced patient sample. Only offspring searching for information contributed to those studies. There wasn’t a practical way to capture the opinions of those offspring who were not part of the Internet support groups, who may indeed represent a very silent majority.
Do embryo donor-conceived offspring have the right to know their origins?
Many donor-conceived individuals feel they have a right to know their genetic origins (Shehab D, et al. 2008). Embryo donors and recipients, who may prefer an anonymous process, have rights that compete with those of the donor-conceived individuals. Embryo donors and recipients have legal rights that are in direct contrast to the offspring’s moral view that family secrets shouldn’t be kept. There is no easy way to reconcile these contrasting legal rights and moral perspectives. Most agree, at the risk of offending those who feel differently, that in a society ruled by law, a legal right ultimately trumps a perceived moral right. Thus, the legal rights of donors and recipients continue to prevail in this country.
Should disclosure be mandated?
There is a stronger preference to discard or abandon the embryos than there is to donate them. Placing any impediments on embryo donation, such as mandating disclosure, may not only reduce the number of embryos donated but will almost certainly result in a greater number of embryos discarded or abandoned. I cannot imagine the well-meaning individuals who are asking for mandated disclosure would want the loss of embryos and potential families to be an almost certain unintended consequence.
Parents who used donor material feel the decision to disclose is private, highly personal and should be left to the discretion of the individual families and not regulated in any way (Shehab D, et al. 2008). A dual-track of disclosure/nondisclosure system will most meet the needs of everyone (De Jonge C, et al. 2006). We need to educate, guide and support, but not force these processes.
What happens to children who learn or don’t learn about their origins?
Interestingly, children do not seem to be harmed if disclosure does not take place.
Warm parent-child relationships and positive child development have been documented, although few of these donor embryo-conceived children have yet entered adolescence (Golombok S, et al. 2006). While more data and follow up studies are certainly needed, embryo donation children do not seem to be at increased risk for developing psychological problems during early and middle childhood, regardless of disclosure decisions. Supporting these findings about early disclosure, neither the children nor the family seem to be harmed (MacCallum F, et al. 2007 & 2008). While not definitive, the research we have so far suggests that disclosure decisions, either for or against, will not cause irreparable harm to the embryo donor-conceived individuals or the parents who raised them.
Interestingly, research shows that embryo donation families are more child-centered than adoptive and, even, other IVF families, regardless of their disclosure status (MacCallum F, et al. 2007 & 2008). This may be due in part to the recipients’ older age and maturity compared to younger IVF and adoptive parents. Embryo donation-conceived children are tremendously appreciated, greatly desired and hard fought for by their parents. This should give embryo donors confidence that donor-conceived offspring will be well cared for by wonderful recipient families.
What should be considered if embryo donor-conceived individuals meet the donors?
If donor-conceived individuals connect their donors and/or blood siblings, it is important that everyone’s expectations be reasonable. Members of donor-linked families may have a significant mismatch regarding their levels of hope and expectation about contact (Scheib JE, et al. 2008). Some of the participants may have a tendency to romanticize the first meeting and future relationship. The participants should move slowly and carefully, without inappropriate expectations.
It is exceedingly important that the relationship between the parents who raised the child and the embryo donor-conceived offspring not be harmed, as this is the basis for the child’s stability and home life. The fear that this relationship will be damaged is one of the main reasons recipients prefer to not disclose, so it must be protected.
Long-term data regarding the relationships between embryo donors and embryo donor-conceived individuals is not yet available, so we simply are unable to tell any of the participants what the outcome may be (Grotevant HD, et al, 2008). The parents who raised the donor-conceived child may feel threatened by the relationship between the donors and offspring. It would be irresponsible for us as clinicians to assume that all will go well. We must remain cautious and counsel our patients carefully and individually until we have more information to guide them.
Can recipients get help in deciding if disclosure should take place?
Consultation with a mental health professional (MHP) regarding disclosure is strongly encouraged but the counselor must understand the subtle differences between embryo donation and adoption, where disclosure is the norm. Neutrality is required and finding a skilled and experienced MHP is important. Many patients resent direct suggestions about disclosing and far prefer a discussion that examines their own needs and perspectives (Klock SC, 1997). In reality, it is really not appropriate to give a uniform recommendation that does not take into account the personal, ethical and religious views of the embryo recipients.
Even more important, embryo recipients may want to hear from other recipients, people who have worked through the issues or are struggling with making the decision (Klock SC, 1997).
In our three recent surveys examining the perspectives of potential embryo donors, recipients and offspring, there were a few consistent findings:
- From the embryo donor perspective, the majority polled desired an open process.
- From the embryo recipient perspective, the majority polled desired anonymity.
- The majority of potential embryo donors and embryo recipients would disclose to their friends and family.
- If open-identity was available, the majority felt information should be shared and potential contact be made before the age of 18, which may differ in adoption proceedings.
Are reproductive facilities prepared for disclosure?
A medical providers’ first reaction will be to protect their patients: the embryo donors and recipients. State and federal statutes protect confidentiality and clear consent must always be provided before medical/identifying information is released to a third party. The reproductive facilities also will want to be insulated from any legal consequences should disclosure occur.
Some medical practices will be operationally challenged to have records available years after procedures are performed. Many states require that medical records be retained for seven years. The Federal Drug Administration (FDA) requires practices to retain for ten years the medical records of patients involved in third party conception, while the ASRM suggests the records be stored indefinitely. Being able to identify those particular charts from others that would otherwise routinely be discarded is a logistical challenge. In my practice, we color-code the charts involving any type of donor material, making them easy to pull and save.
I also feel embryo recipients have a responsibility to keep the information they were originally provided about their embryo donors protected and safe in case the practice’s medical records are ever lost or deleted.
Regardless, the facilities have a responsibility to assure charts are available and that systems are in place to make disclosure possible. Policies and procedures will need to be created so that they are ready when the requests come. Mirroring the adoption world in this instance may be of use.
How do recipients disclose?
Experienced MHP’s can provide invaluable assistance in helping embryo recipients present this  sensitive information. Reproductive endocrinologists, well versed in this topic, may also be very useful in deciding how and when to tell the children. Forums where parents can discuss these issues with other parents who have lived them also may be indispensible. While few books exist on the topic, I will be reviewing one quite soon that does. Please stay tuned.
sensitive information. Reproductive endocrinologists, well versed in this topic, may also be very useful in deciding how and when to tell the children. Forums where parents can discuss these issues with other parents who have lived them also may be indispensible. While few books exist on the topic, I will be reviewing one quite soon that does. Please stay tuned.
Using the following phrases and ideas in the discussion may be useful:
- We wanted you to be in our lives so much, we had to travel far and wide to find you.
- The people who gave us their embryos, which included you, gave us the most wonderful gift in the world. They loved you so much that they wanted you to enjoy life with us.
- I got to carry and protect you, giving you all the love I could.
- Remember that eggs, sperm and embryos don’t make a family any more than bricks make a happy home. What makes a family is how we support and love each other.
A good way to start this conversation is by emphasizing love, your commitment to find them, the amount of time, effort and emotion you invested in bringing them into the world, and the fact that genetics doesn’t necessarily make a family. I’m sure MHP’s will offer recipients some great phrases and ideas, which I will encourage that they share with us on this blog.
What are the long-term consequences of disclosure?
Research is needed to find out more about long-term disclosure outcomes. It is difficult to ascertain how children are fairing who are not told about their origins fare because these individuals are not readily accessible to research. Also, what happens over time with open donation is largely unknown. I feel it is unrealistic to assume that everyone will live together as one big happy family. I suspect, however, that the donor-conceived children will benefit in establishing connections with blood siblings.
If disclosure occurs and the offspring are destined to contact their genetic parents, their reunion should not be romanticized. They will not necessarily be welcomed with open arms up to two decades later by the donors. In fact, we don’t have any idea how well or how disruptive these reunions will be. Once again, research is desperately needed.
Is there a disclosure compromise available?
The idea of open-identity is a fascinating concept for embryo donation. This would allow donors to be contacted if the recipients eventually disclose the genetic origins to their donor-conceived offspring. According to our recent surveys, most respondents wanted disclosure with open-identity to occur before the children turn 18 and have the ability to contact the donors. But what harm will come to the child if disclosure and contact are done earlier in the formative years? Would there be some advantages to knowing earlier rather than later in adulthood?
It would seem that if recipients are disclosing for the sake of the child, it may truly be best to allow disclosure and open-identity earlier rather than later. I hope to pose this question to MHPs in the months to come.
In summary
I feel it is far better that we design programs with open-identity options but not legislate them. Legislation, especially if it is retroactive, will inject uncertainty about every current rule governing the practice of egg/sperm/embryo donation (Guido P, 2001). Legislation will do more to shut donation processes down than any other development. I understand there are some donor-conceived individuals who feel that all information should be open to review, but past contracts were doubtfully formulated that way. Although unpopular with some, the perceived rights of the donor offspring should not be allowed to circumvent the true legal rights of the donors and recipients.
At a recent ASRM meeting, some MHPs and reproductive attorneys lamented that anonymous embryo donations are allowed to continue. I explained that if they want to change physicians’ and patients’ perspectives on anonymity, they would only succeed with data showing the long-term benefits of disclosure. Until that time, clinicians and patients alike will be hesitant to adopt a stance that is not yet supported by unbiased research. We should tread lightly and make very few assumptions when it comes to issues so important as disclosure or we risk doing harm to our patients. We all need a bit more information in caring for our embryo donors and recipients. We must never forget, however, that embryo donation-conceived individuals, who were never our actual patients, and are still owed thoughtful care and tremendous compassion. We must keep an open mind and learn from their perspective.
There will always be a group of patients who will want anonymity and we all need to respect their wishes. Everyone involved in this debate needs to take a breath and step back. I grow weary of a very vocal group of patients who believe their way is the single right way. This is not a black and white issue. We should instead see the disclosure controversy in shades of grey. Compromise these days seems to be viewed as form of weakness in our country’s politics. This country was founded on the basis of choice and I feel strongly that we should design systems that will allow for choice while educating the participants of embryo donation about their options and the potential results of both disclosure and non-disclosure to embryo donor-conceived individuals. It is our responsibility.
As always I welcome a respectful dialogue, especially from those who disagree. I look forward to reviewing research studies that will guide all of us so we may better counsel embryo donors, embryo donor recipients and especially the embryo donor-conceived individuals, who remain the final focus of this entire discussion.
Craig R. Sweet, M.D.
Reproductive Endocrinologist
Founder, Medical & Practice Director
Embryo Donation International
Info@EmbryoDonation.com
www.EmbryoDonation.com
Associated Blog Segments:
- Introduction: Disclosure Issues in Embryo Donation
- Embryo Donor Perspective
- Survey Results: Imagine you are an Embryo Donor
- Blog: Disclosure Issues From the Perspective of the Embryo Donor (Part 1 & Part 2)
- Embryo Recipient Perspective
- Survey Results: Imagine you are an Embryo Recipient
- Blog: Disclosure Issues From the Perspective of the Embryo Recipient (Part 1 & Part 2)
- Embryo Donor Offspring Perspective
- Survey Results: Imagine you are an Embryo Donor Offspring
- Blog: Disclosure Issues From the Perspective of the Embryo Donor Offspring (Part 1 & Part 2)
Registry Links:
- The Donor Sibling Registry: https://www.donorsiblingregistry.com/
- UK Donor Link: http://ukdonorlink.org.uk/ (funding uncertain)
- Western Australia’s Voluntary Register for Donors, Offspring and their Families: http://www.voluntaryregister.health.wa.gov.au/home/
- BCdonorconception, British Columbia donor offspring: http://groups.yahoo.com/group/BCdonorconception/
Donor Sperm Facility Registries:
- California Cryobank’s Sibling Registry: http://www.sibling-registry.com/
- Fairfax Cryobank: Donor Sibling Groups: http://www.donorsiblinggroups.com/Home_Page.html
General Third Party Conception Support:
- Donor Conception Support Group: http://www.dcsg.org.au/
- PCVAI, People Conceived via Donor Insemination: http://groups.yahoo.com/group/PCVAI/
- International Donor Offspring Alliance: http://www.idoalliance.org/
- The Donor Conception Support Group of Australia Inc: http://www.clan.org.au/dcsg/index.html
- Canadian Donor Offspring: http://www.canadiandonoroffspring.ca/
- Germany: http://www.spenderkinder.de/
References:
Beeson DR, Jennings PK, Kramer W. Offspring searching for their sperm donors: how family type shapes the process. Hum Reprod. 2011 Sep;26(9):2415-24.
Daniels K. The controversy regarding privacy versus disclosure among patients using donor gametes in assisted reproductive technology. J Assist Reprod Genet. 1997 Aug;14(7)-373-5.
De Jonge C, Barratt CL. Gamete donation- a question of anonymity. Fertil Steril. 2006 Feb;85(2)-500-1.
Klock SC. The controversy surrounding privacy or disclosure among donor gamete recipients. J Assist Reprod Genet. 1997 Aug;14(7)-378-80.
Ethics Committee of the American Society for Reproductive Medicine. Informing offspring of their conception by gamete donation. Fertil Steril. 2004 Sep;82 Suppl 1-S212-6.
Ethics Committee of the ASRM. Informing Offspring of their conception by gamete donation. Fertil Steril 2004;81(3):527-31.
Golombok S, Lycett E, MacCallum F, Jadva V, Murray C, Rust J, Abdalla H, Jenkins J, Margara R. Parenting infants conceived by gamete donation. J Fam Psychol. 2004 Sep;18(3):443-52
Golombok S, Murray C, Jadva V, Lycett E, MacCallum F, Rust J. Non-genetic and non-gestational parenthood: consequences for parent-child relationships and the psychological well-being of mothers, fathers and children at age 3. Hum Reprod. 2006 Jul;21(7):1918-24.
Grotevant HD, Wrobel GM, Von Korff L, Skinner B, Newell J, Friese S, McRoy RG. Many Faces of Openness in Adoption: Perspectives of Adopted Adolescents and Their Parents. Adopt Q. 2008 Jul 1;10(3 & 4):79-101.
Guido P. The reduction of sperm donor candidates due to the abolition of the anonymity rule: Analysis of an argument. J Assist Reprod Genet. 2001 Nov;18(11)-617-22.
Jadva V, Freeman T, Kramer W, Golombok S. The experiences of adolescents and adults conceived by sperm donation: comparisons by age of disclosure and family type. Hum Reprod. 2009 Aug;24(8):1909-19.
Klock SC. The controversy surrounding privacy or disclosure among donor gamete recipients. J Assist Reprod Genet. 1997 Aug;14(7)-378-80.pdf
MacCallum F, Golombok S, Brinsden P. Parenting and child development in families with a child conceived through embryo donation. J Fam Psychol. 2007 Jun;21(2):278-87.
MacCallum F, Keeley S. Embryo donation families: a follow-up in middle childhood. J Fam Psychol. 2008 Dec;22(6):799-808.
Mahlstedt PP, LaBounty K, Kennedy WT. The views of adult offspring of sperm donation: essential feedback for the development of ethical guidelines within the practice of assisted reproductive technology in the United States. Fertil Steril. 2010 May 1;93(7):2236-46.
Ravitsky, V. & Scheib, J.E. (2010). Donor-conceived individuals’ right to know. Hastings Center Bioethics Forum 2010;40:(4).
Scheib JE, Ruby A. Contact among families who share the same sperm donor. Fertil Steril. 2008 Jul;90(1):33-43.
Shehab D, Duff J, Pasch LA, Mac Dougall K, Scheib JE, Nachtigall RD. How parents whose children have been conceived with donor gametes make their disclosure decision: contexts, influences, and couple dynamics. Fertil Steril. 2008 Jan;89(1):179-87.
Widdows H, MacCallum F. Disparities in parenting criteria: an exploration of the issues, focusing on adoption and embryo donation. J Med Ethics 2002;28:139-42.